Okayama University Medical Research Updates (OU-MRU) Vol.13
August 05, 2015
Source: Okayama University, Center for Public Affairs and Communications
For immediate release: 05 August 2015
Okayama University research: Peptide directs artificial tissue growth
(Okayama, 05 August) Studies on peptide-modified cultures demonstrate control over both tissue growth and location in vitro.
“In vitro fabricated biological tissue would be a valuable tool to screen newly synthesized drugs or understand the tissue development process,” explain Takuya Matsumoto and his colleagues in a recent report. However tissues grown so far have not reached the size and final shape of natural tissue. Matsumoto and colleagues at Okayama, Osaka and Kansai Universities in Japan have now identified a tripeptide sequence that can be used to overcome both limitations. With hydrogel modified with the tripeptide they demonstrate enhanced tissue growth of a salivary gland both across a sheet of tissue and at specified localised points.
The salivary gland studied - the submandibular gland - develops a branched structure through budding and the elongation of ducts, which is also typical of other structures such as lungs, kidneys, mammary glands and spleen. Previous research revealed the importance of the glycoprotein fibronectin in the morphological development and growth of salivary gland tissue. Consequently Matsumoto and colleagues focused their investigations on the effects of the specific tripeptide sequence arginine-glycine aspartic acid (RGD) that acts as a cell binding site on fibronectin.
The researchers studied the growth of salivary gland tissue on hydrogel both modified with different concentrations of RGD and without and found significant growth enhancements that increased with RGD concentrations. They then experimented with RGD-modified hydrogel beads and demonstrated that localised growth enhancement occurred where the beads were situated, which could provide control over morphology in artificial tissue growth. Further studies also suggested that neuronal growth and the enhancement of growth factor proteins were responsible for the enhanced gland tissue growth observed.
In their report of the work the researchers conclude, “This in vitro SMG tissue growth modulation system can have a variety of uses including tissue arrays for drug screening and as a biologic tool to understand tissue development.”
Background
Tripeptide in previous studies
The shape development or morphogenesis of the submandibular gland develops through the formation of clefts that occur through a change in cell adhesion so that they bind to the surrounding extracellular matrix instead of each other. Previous research has shown that expression of the glycoprotein fibronectin is crucial for cleft formation.
Integrin is a receptor protein that bridges interactions between cells and the extracellular matrix. The domain of fibronectin that binds to integrin is the tripeptide sequence arginine-glycine aspartic acid (RGD) prompting studies of this tripeptide sequence on tissue growth.
Investigations have already shown that transplanting composites containing RGD peptides onto the back of a mouse can result in the development of growth plates – the tissue at the ends of bones in growing children and adolescents. However so far there have been no studies into the growth and morphology of tissues in vitro. The ability to grow tissues in vitro is particularly important for drug screening and studies to better understand tissue growth.
The role of FGFs
Fibroblast growth factors (FGFs) are a family of proteins involved in embryonic development, wound healing and signalling pathways. Two specific FGFs play a crucial role in the growth of the two main tissue types in the submandible salivary gland – FGF7 induces budding in the epithelial tissue and FGF10 induces the elongation of ducts in the mesenchymal tissue.
Matsumoto and colleagues imaged the salivary gland tissues grown in RGD-modified hydrogels using immunofluorescent staining and found both FGF7 and FGF10 were highly expressed. Experiments to grow the salivary gland tissues in the presence of FGF7 and FGF10 antibodies revealed that these antibodies inhibited tissue growth. The RGD-modified hydrogel was also ineffective at enhancing tissue budding in isolated epithelial cells alone.
The role of neurites
Previous studies have also suggested that neuron outgrowths – ‘neurites’ – from a region known as the parasympathetic ganglion in submandibular gland tissue are key to tissue growth. The researchers observed an even distribution of the neurite network throughout the gland tissues they had grown. Antibodies to the neurotrophic factors, which are responsible for neuron growth and survival, also inhibited submandibular gland tissue growth in RGD-modified hydrogels.
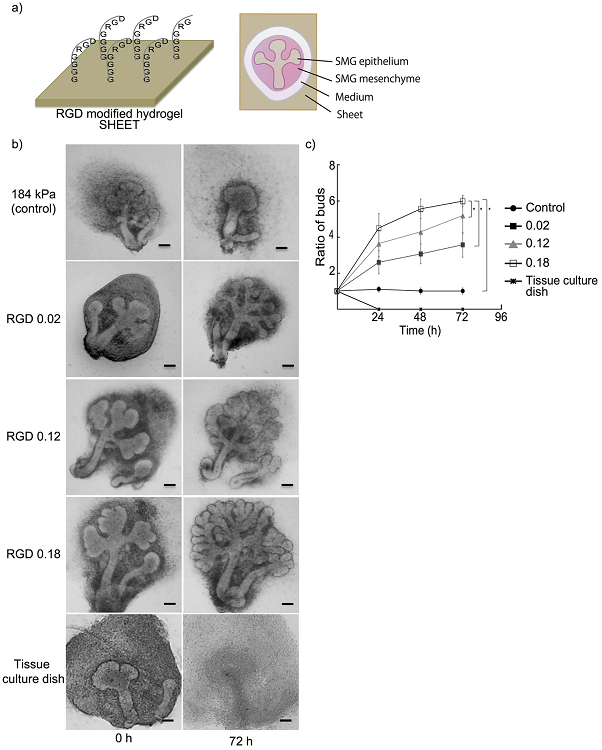
Figure caption
a) Schematic illustration of the RGD-modified hydrogel (left) and the submandibular gland (SMG) tissue culture on hydrogel (right). b,c) SMG cultured on hydrogel sheets modified by introducing various RGD concentrations (0–0.18 mol/l), and SMG cultured on a tissue culture dish without any hydrogel substrate as a negative control (Bar = 50 μ m). A stiffer gel sheet normally attenuates SMG growth, but the SMG growth was enhanced when RGD was introduced. The SMG growth changed in accordance with the introduced amount of RGD. SMG was completely dissociated when cultured on the traditional tissue culture dish instead of hydrogel.
Reference
Hiroaki Taketa, Gulsan Ara Sathi, Mahmoud Farahat, Kazi Anisur Rahman, Takayoshi Sakai, Yoshiaki Hirano, Takuo Kuboki, Yasuhiro Torii, Takuya Matsumoto. Peptide-modified Substrate for Modulating Gland Tissue Growth and Morphology In Vitro. Scientific Reports 5, Article number: 11468 (2015).
DOI: 10.1038/srep11468
http://www.nature.com/articles/srep11468
Correspondence to
Professor Takuya Matsumoto, D.D.S., Ph.D.
Department of Biomaterials, Okayama University,
2-5-1 Shikata-Cho, Okayama, 700-8558, Japan.
E-mail: [email protected]
Contact information
Public Relations and Information Strategy
E-mail: www-adm(a)adm.okayama-u.ac.jp
For inquiries, please contact us by replacing (a) with the @ mark.
Website: //www.okayama-u.ac.jp/index_e.html
Okayama Univ. e-Bulletin: //www.okayama-u.ac.jp/user/kouhou/ebulletin/
Okayama Univ. e-Bulletin (PDF Issues): //www.okayama-u.ac.jp/user/kouhou/ebulletin/ebulletin.html
About Okayama University (You Tube): https://www.youtube.com/watch?v=iDL1coqPRYI
Okayama University Image Movie (You Tube): https://www.youtube.com/watch?v=_WnbJVk2elA
Okayama University Medical Research Updates (OU-MRU)
Vol.1:Innovative non-invasive ‘liquid biopsy’ method to capture circulating tumor cells from blood samples for genetic testingVol.2:Ensuring a cool recovery from cardiac arrestVol.3:Organ regeneration research leaps forwardVol.4:Cardiac mechanosensitive integratorVol.5:Cell injections get to the heart of congenital defectsVol.6:Fourth key molecule identified in bone developmentVol.7:Anticancer virus solution provides an alternative to surgeryVol.8:Light-responsive dye stimulates sight in genetically blind patientsVol.9:Diabetes drug helps towards immunity against cancerVol.10:Enzyme-inhibitors treat drug-resistant epilepsyVol.11:Compound-protein combination shows promise for arthritis treatmentVol.12:Molecular features of the circadian clock system in fruit flies
About Okayama University
Okayama University is one of the largestcomprehensive universities in Japan with roots going back to the Medical Training Place sponsored by the Lord of Okayama and established in 1870. Now with 1,300 faculty and 14,000 students, the University offers courses in specialties ranging from medicine and pharmacy to humanities and physical sciences. Okayama University is located in the heart of Japan approximately 3 hours west of Tokyo by Shinkansen.